Robert M. Friedlander, MD, MA
Dr. Robert Friedlander is no longer affiliated with UPMC or the University of Pittsburgh, stepping down from his role as Chair of the University of Pittsburgh Department of Neurological Surgery and as co-director of the UPMC Neurological Institute in December of 2025.
Dr. Friedlander's research focused on synthesis of melatonin in the brain and if the natural decline in melatonin synthesis with age contributed to age-related neurodegeneration. A breakthrough in this regard would lead to the establishment of a mouse model of age-related neurodegeneration. Additionally, a future goal of this model would be its use to identify new strategies for preventing neuro-degenerative diseases, such as dementia.
His major research interests lie in the study of the mechanistic pathways of the caspase apoptosis gene family (cell death). His work includes the evaluation of treatment strategies for neurodegenerative diseases (Huntington’s and ALS), stroke, brain trauma, and spinal cord injury through the modulation of the caspase-family apoptotic pathways.
"Dementia robs people from their mind and what are we without our thoughts, what are we without being able to talk? All of these issues, are what we are trying to address."
- Dr. Robert Friedlander
Explore Friedlander's Dementia Research
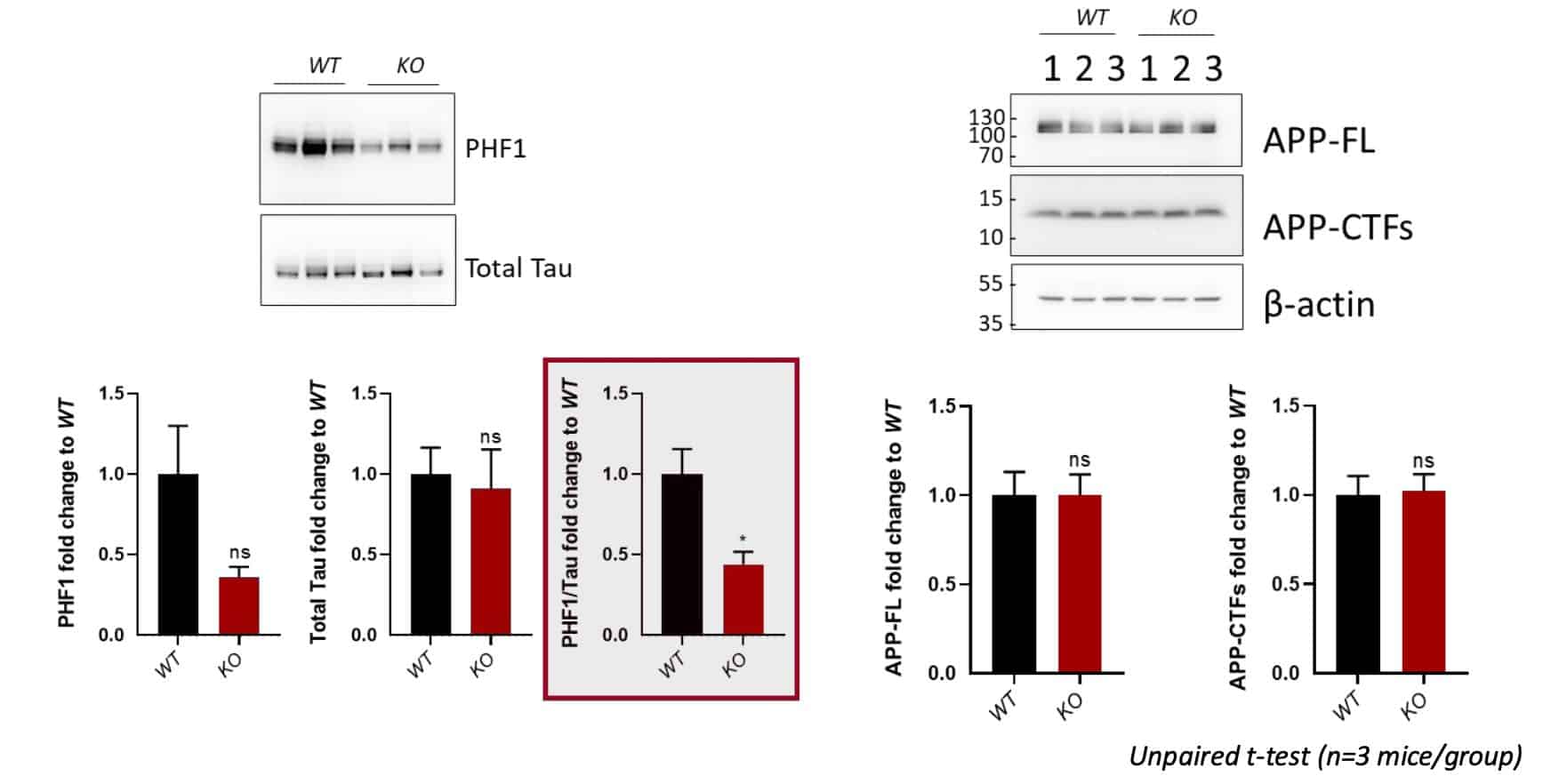
Dr. Robert M. Friedlander and his team have continued to pursue research to understand the role of melatonin in inhibiting neuroinflammation and slowing dementia in a preclinical mouse model. Research has shown that melatonin levels deplete severely with aging and more so in patients with Alzheimer’s disease (AD). Support from the CTF Consortium led to the establishment of a novel mouse model of age-related neurodegeneration genetically which was engineered not to make melatonin (AANAT-KO). This mouse model in tandem with Dr. Thathiah’s lab, has been used to better understand the accumulation of phosphorylated tau, which causes synaptic impairment and neuronal dysfunction - a key pathological feature of Alzheimer's disease.
Findings:
- Confirmed phosphorylated tau levels are reduced in melatonin-deficient mice (AANAT KO mice)
- *Amyloid precursor protein (APP) levels are unchanged in melatonin-deficient mice –Aβ levels have not yet been checked.
- Confirmed no APP gene variant changes and validates use of the AANAT KO mice for further studies.
- Tau phosphorylation signature can differ - determination of the status of tau phosphorylation at additional sites will provide insight into the implications of reduced phosphorylation at the PHF1 site for disease.
*Amyloid precursor protein (APP) is abundant in neurons and gives rise to amyloid-β (Aβ) peptides
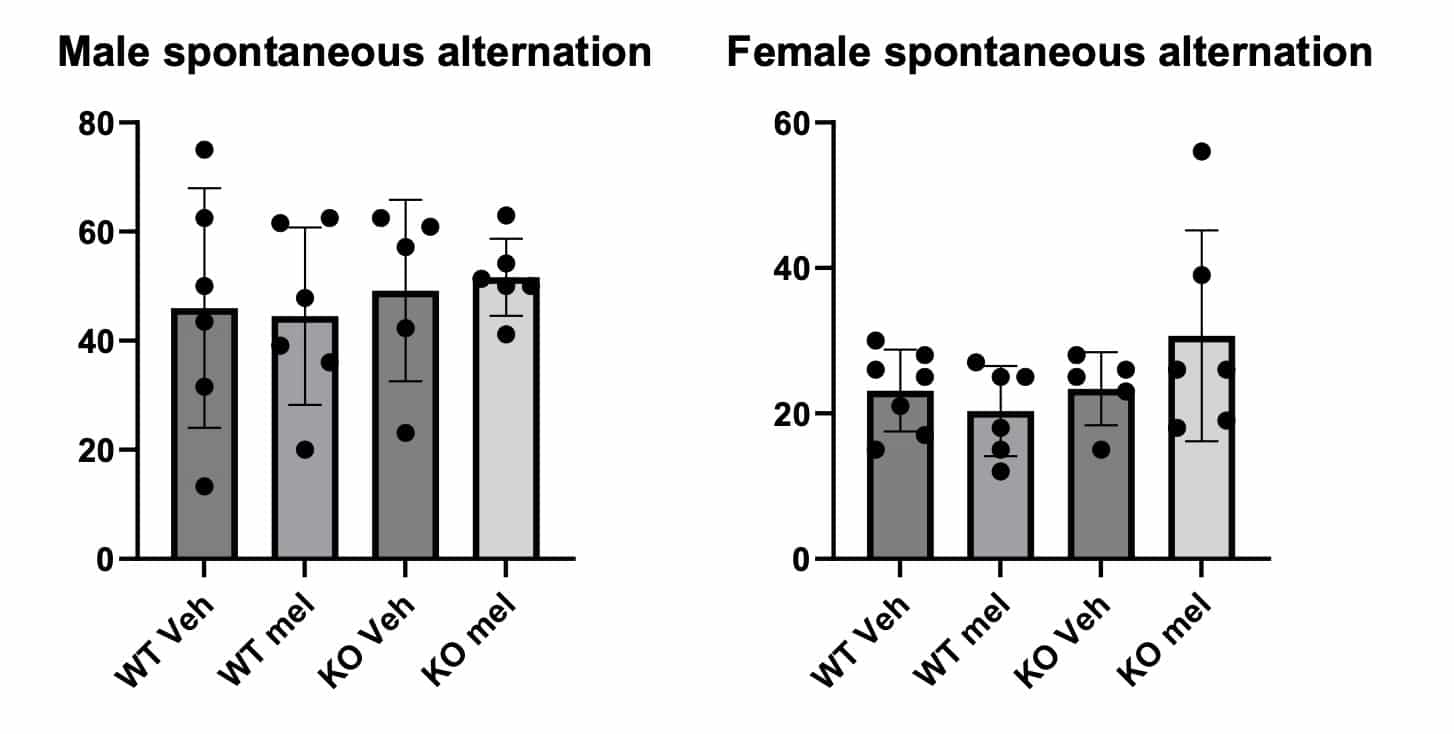
Dementia is known to develop with different frequency in males and females, depending upon the disease form. Dr. Friedlander’s lab has begun the necessary research to better understand these differences with melatonins impact on memory and brain volume. Preliminary data showed that female AANAT-KO mice had memory deficits at 1 year old compared with WT mice and male AANAT-KO mice had decreased brain volume at 1 year old compared with WT mice.
Upon continued testing, at 12 months of age with 6 months of melatonin supplementation, the average memory score for AANAT- KO mice was higher in both males and females, but due to the large variability within the groups, this difference was not statistically significant. Given this factor, brain tissue samples will be collected with future funding at 24 months of age, NIH’s standard for an aged mouse equivalent to an elderly human. Once brain tissue samples can be collected, MRI images will be used to calculate brain and hippocampal volumes, creating a better indication of melatonin’s impact as a dementia therapeutic for both men and women.
Additional research findings built upon these discoveries can be found under Dr. Amantha Thathiah's research as their labs worked closely on these projects to determine outcomes.
In 2022 and 2023, Dr. Friedlander’s lab submitted two NIH applications for large scale research projects to expand upon specific aims in relation to the funded research of the CTF Consortium. This is reason your continued funding is vitally important to help secure the necessary preliminary data needed for future awarded grant funding from the NIH. NIH application details are below:
2022 NIH Submission
Requested Support: $2.1 million dollars
Research Duration: 5 years
Project Title: Melatonin deficits cause mitochondrial biomass reduction in age-related neurodegeneration
Synopsis: This project aims to investigate the role of mitochondrial homeostasis in maintaining brain health, and the potential link between melatonin levels and mitochondrial homeostasis.
2023 NIH Submission
Requested Support: $3.5 million dollars
Research Duration: 5 years
Project Title: Mechanisms of Melatonin deficiency in age-related neurodegeneration
Synopsis: This project proposes a longitudinal study in newly developed melatonin deficient mice to determine melatonin causality in neurodegeneration, as well as test oral melatonin supplementation to prevent age-related cognitive decline and neurodegeneration.
